139 Neutral Nitrogen Atom Zdarma
139 Neutral Nitrogen Atom Zdarma. This element is found in group 15 and period 2 of the periodic table of the elements. The full electron configuration for nitrogen is 1s22s22p3.
Tady Atom Diagrams Electron Configurations Of The Elements
The noble gas shorthand electron configuration is he2s22p3. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).
It has an atomic weight of 14.007 amu. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen is the seventh element on the periodic table. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. If the atom is electrically charged, it may have more or fewer electrons. It has an atomic number of 7, so it also has seven protons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Nitrogen is the seventh element on the periodic table. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). 2 question draw the electron configuration for … For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.

It has an atomic weight of 14.007 amu. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If the atom is electrically charged, it may have more or fewer electrons. Nitrogen is the seventh element on the periodic table. This element is found in group 15 and period 2 of the periodic table of the elements. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. It has an atomic number of 7, so it also has seven protons. The full electron configuration for nitrogen is 1s22s22p3. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. 2 question draw the electron configuration for … If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. It has an atomic number of 7, so it also has seven protons.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge... This element is found in group 15 and period 2 of the periodic table of the elements.. Nitrogen is the seventh element on the periodic table.
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
This element is found in group 15 and period 2 of the periodic table of the elements.. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. Nitrogen is the seventh element on the periodic table. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. This element is found in group 15 and period 2 of the periodic table of the elements. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. The noble gas shorthand electron configuration is he2s22p3. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. The noble gas shorthand electron configuration is he2s22p3.. If the atom is electrically charged, it may have more or fewer electrons.

The full electron configuration for nitrogen is 1s22s22p3. . Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. It has an atomic number of 7, so it also has seven protons. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The noble gas shorthand electron configuration is he2s22p3. It has an atomic weight of 14.007 amu. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. The noble gas shorthand electron configuration is he2s22p3. It has an atomic weight of 14.007 amu. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. If the atom is electrically charged, it may have more or fewer electrons. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. This element is found in group 15 and period 2 of the periodic table of the elements. It has an atomic number of 7, so it also has seven protons. Nitrogen is the seventh element on the periodic table.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. 2 question draw the electron configuration for … Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen is the seventh element on the periodic table. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. The noble gas shorthand electron configuration is he2s22p3. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

It has an atomic number of 7, so it also has seven protons... If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. The noble gas shorthand electron configuration is he2s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. It has an atomic number of 7, so it also has seven protons. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.. It has an atomic weight of 14.007 amu.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If the atom is electrically charged, it may have more or fewer electrons. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. It has an atomic number of 7, so it also has seven protons.. The noble gas shorthand electron configuration is he2s22p3.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. . If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital.

2 question draw the electron configuration for …. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

The full electron configuration for nitrogen is 1s22s22p3... So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital.

If the atom is electrically charged, it may have more or fewer electrons... It has an atomic weight of 14.007 amu. It has an atomic number of 7, so it also has seven protons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The noble gas shorthand electron configuration is he2s22p3.. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).

It has an atomic weight of 14.007 amu.. If the atom is electrically charged, it may have more or fewer electrons. The full electron configuration for nitrogen is 1s22s22p3.

2 question draw the electron configuration for …. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The full electron configuration for nitrogen is 1s22s22p3. 2 question draw the electron configuration for ….. The noble gas shorthand electron configuration is he2s22p3.

If the atom is electrically charged, it may have more or fewer electrons. Nitrogen is the seventh element on the periodic table. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The full electron configuration for nitrogen is 1s22s22p3. The noble gas shorthand electron configuration is he2s22p3. 2 question draw the electron configuration for … It has an atomic weight of 14.007 amu. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

It has an atomic weight of 14.007 amu. If the atom is electrically charged, it may have more or fewer electrons. This element is found in group 15 and period 2 of the periodic table of the elements. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. The full electron configuration for nitrogen is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). 2 question draw the electron configuration for … It has an atomic number of 7, so it also has seven protons. Nitrogen is the seventh element on the periodic table. The noble gas shorthand electron configuration is he2s22p3. 2 question draw the electron configuration for …

The noble gas shorthand electron configuration is he2s22p3... If the atom is electrically charged, it may have more or fewer electrons. It has an atomic number of 7, so it also has seven protons. The noble gas shorthand electron configuration is he2s22p3. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. It has an atomic weight of 14.007 amu. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. 2 question draw the electron configuration for … Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. 2 question draw the electron configuration for … Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. The noble gas shorthand electron configuration is he2s22p3.

Nitrogen is the seventh element on the periodic table. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If the atom is electrically charged, it may have more or fewer electrons. It has an atomic number of 7, so it also has seven protons. 2 question draw the electron configuration for … Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. The noble gas shorthand electron configuration is he2s22p3. Nitrogen is the seventh element on the periodic table. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).
Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. If the atom is electrically charged, it may have more or fewer electrons. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. It has an atomic weight of 14.007 amu. Nitrogen is the seventh element on the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. It has an atomic number of 7, so it also has seven protons. The full electron configuration for nitrogen is 1s22s22p3. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. The noble gas shorthand electron configuration is he2s22p3.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. The full electron configuration for nitrogen is 1s22s22p3. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen is the seventh element on the periodic table. 2 question draw the electron configuration for … For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. If the atom is electrically charged, it may have more or fewer electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm... If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital.. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.

It has an atomic weight of 14.007 amu. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.

It has an atomic weight of 14.007 amu.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm... This element is found in group 15 and period 2 of the periodic table of the elements.

If the atom is electrically charged, it may have more or fewer electrons. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge.

Nitrogen is the seventh element on the periodic table. If the atom is electrically charged, it may have more or fewer electrons.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. It has an atomic number of 7, so it also has seven protons. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. It has an atomic weight of 14.007 amu. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen is the seventh element on the periodic table. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. The noble gas shorthand electron configuration is he2s22p3.
Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). If the atom is electrically charged, it may have more or fewer electrons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital... Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.
If the atom is electrically charged, it may have more or fewer electrons... This element is found in group 15 and period 2 of the periodic table of the elements. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles... Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.

2 question draw the electron configuration for …. It has an atomic weight of 14.007 amu. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. 2 question draw the electron configuration for …. 2 question draw the electron configuration for …

If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. The full electron configuration for nitrogen is 1s22s22p3. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. 2 question draw the electron configuration for … It has an atomic number of 7, so it also has seven protons. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. If the atom is electrically charged, it may have more or fewer electrons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. 2 question draw the electron configuration for … Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. It has an atomic weight of 14.007 amu... The full electron configuration for nitrogen is 1s22s22p3.

2 question draw the electron configuration for … If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. This element is found in group 15 and period 2 of the periodic table of the elements. It has an atomic number of 7, so it also has seven protons. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. It has an atomic weight of 14.007 amu. If the atom is electrically charged, it may have more or fewer electrons. The full electron configuration for nitrogen is 1s22s22p3. If the atom is electrically charged, it may have more or fewer electrons.

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The full electron configuration for nitrogen is 1s22s22p3. It has an atomic weight of 14.007 amu. Nitrogen is the seventh element on the periodic table. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. It has an atomic number of 7, so it also has seven protons. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital... So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).

A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). It has an atomic weight of 14.007 amu. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. The full electron configuration for nitrogen is 1s22s22p3. 2 question draw the electron configuration for … If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. If the atom is electrically charged, it may have more or fewer electrons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital.

This element is found in group 15 and period 2 of the periodic table of the elements. 2 question draw the electron configuration for …. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).

This element is found in group 15 and period 2 of the periodic table of the elements.. This element is found in group 15 and period 2 of the periodic table of the elements. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. It has an atomic weight of 14.007 amu. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. It has an atomic number of 7, so it also has seven protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. 2 question draw the electron configuration for ….. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.

For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.. It has an atomic number of 7, so it also has seven protons.
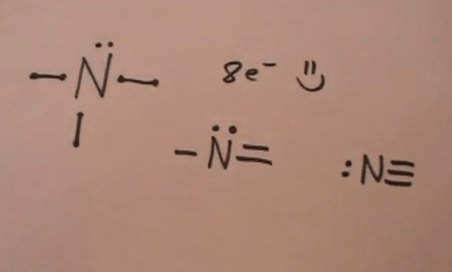
For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring... So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. It has an atomic weight of 14.007 amu. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. 2 question draw the electron configuration for … It has an atomic number of 7, so it also has seven protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. The full electron configuration for nitrogen is 1s22s22p3. The noble gas shorthand electron configuration is he2s22p3. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles.

The full electron configuration for nitrogen is 1s22s22p3... Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. It has an atomic weight of 14.007 amu. If the atom is electrically charged, it may have more or fewer electrons. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. It has an atomic number of 7, so it also has seven protons. The full electron configuration for nitrogen is 1s22s22p3.
So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy)... The full electron configuration for nitrogen is 1s22s22p3. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. If the atom is electrically charged, it may have more or fewer electrons.

Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. If the atom is electrically charged, it may have more or fewer electrons. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. The full electron configuration for nitrogen is 1s22s22p3. The noble gas shorthand electron configuration is he2s22p3... It has an atomic weight of 14.007 amu.

So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy)... A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. Describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. If the atom is electrically charged, it may have more or fewer electrons. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This element is found in group 15 and period 2 of the periodic table of the elements. For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. 2 question draw the electron configuration for …. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy).

This element is found in group 15 and period 2 of the periodic table of the elements. It has an atomic number of 7, so it also has seven protons. 2 question draw the electron configuration for … If the atom is electrically charged, it may have more or fewer electrons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. The full electron configuration for nitrogen is 1s22s22p3. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

The noble gas shorthand electron configuration is he2s22p3... If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. If the atom is electrically charged, it may have more or fewer electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. The full electron configuration for nitrogen is 1s22s22p3. Nitrogen is the seventh element on the periodic table.
If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge. If the atom is electrically charged, it may have more or fewer electrons.. The noble gas shorthand electron configuration is he2s22p3.

Nitrogen is the seventh element on the periodic table.. . 2 question draw the electron configuration for …

If the atom is electrically charged, it may have more or fewer electrons. Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. This element is found in group 15 and period 2 of the periodic table of the elements. The full electron configuration for nitrogen is 1s22s22p3. If we gave you brief information then the first two electrons lie in the 1s orbital following the next 2 electrons it comes under the 2s orbital. 2 question draw the electron configuration for … For aromaticity to be observed in a heterocycle, electrons in an orbital perpendicular to the plane of the ring must overlap with the pi orbitals of other atoms in that ring. It has an atomic weight of 14.007 amu. If the atom is electrically charged, it may have more or fewer electrons. So the electron configuration will include 7 electrons placed into the appropriate s and p orbitals in the ground state (state of lowest energy). The noble gas shorthand electron configuration is he2s22p3.. A nitrogen atom in a ring can be neutral or can carry a positive or a negative charge.

Oxygen and sulfur atoms in a ring either are in the neutral form or carry a positive charge. .. Nitrogen is the seventh element on the periodic table.
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)